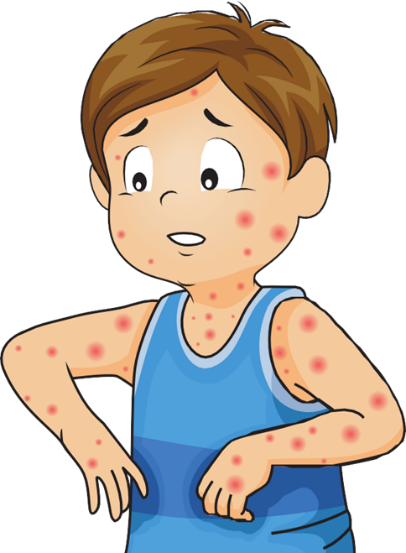
Chickenpox usually begins with:
- Fever
- Tiredness
- Headache
- Loss of appetite
After these initial symptoms, an itchy rash with fluid-filled blisters appears throughout the body. Chickenpox blisters typically start at the chest, back, and face before spreading over the entire body. Blisters usually turn into scabs in five to seven days, after other signs of illness.
Chickenpox can cause several health complications. Complications include skin and soft tissue infections, swelling of the brain (encephalitis), lung infection (pneumonia), bleeding problems, bloodstream infections (sepsis) and dehydration.
Those who are more likely to have serious complications due to chickenpox are:
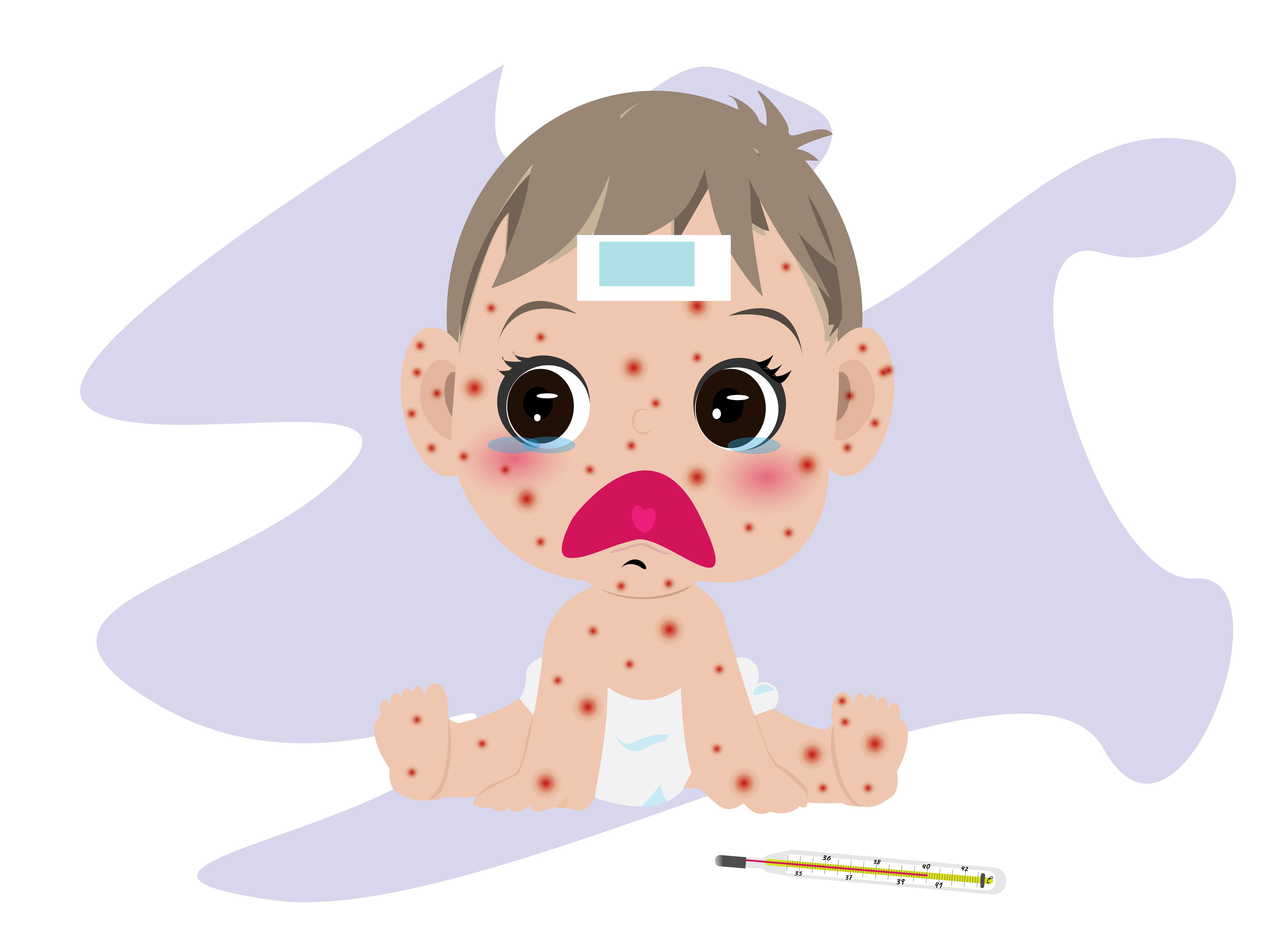
- Infants
- Adolescents
- Adults
- Pregnant people
- People with weakened immune systems
If you think you or someone in your family may have symptoms or have been exposed to chickenpox, talk to your doctor.