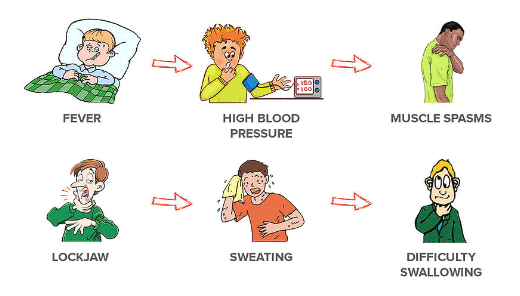
The American Academy of Pediatrics (AAP) and the American Academy of Family Physicians (AAFP) recommend routine tetanus vaccination across the lifespan. Children younger than 7 years of age should receive DTaP (diphtheria, tetanus, acellular pertussis), while older children and adults should receive Tdap (tetanus, diphtheria, acellular pertussis) or Td (tetanus and diphtheria). See the table below for a list of tetanus toxoid-containing vaccines and the recommended schedules.
Multiple combination vaccines containing tetanus toxoid are available for use. Use of combination vaccines are also useful in reducing the number of injections in a single visit.
Tetanus disease may not confer immunity. Unvaccinated or under-vaccinated persons recovering from tetanus should begin or complete their immunization series with tetanus toxoid during convalescence.
Adults
- Administer a single dose of Tdap to adults who have never received Tdap regardless of when they last received Td. This should be followed by a Tdap booster every 10 years.
Catch-Up Guidance for Children 4 Months through 18 Years
- Review the catch-up guidance for diphtheria, tetanus, and pertussis vaccination for children and adolescents whose vaccinations have been delayed.
Guidance for Td
- TdVaxTM has been discontinued and is no longer available in the U.S.
- A limited supply of TENIVAC® (Td) is still available, but due to constraints in vaccine supplies, vaccine providers should follow this guidance until the period of temporary ordering controls for Td vaccine ends:
- Use Tdap vaccine in lieu of Td vaccine whenever possible while Td vaccine supplies are constrained.
- Tdap vaccine is an acceptable alternative to Td vaccine, including when a tetanus booster is indicated for wound management.
- Tdap vaccine isn't an acceptable alternative only when a person has a specific contraindication to pertussis-containing vaccines. Contraindications to DTaP and Tdap occur only very rarely, and in questioning about them, providers should probe whether a true contraindication actually exists. DTaP and Tdap contraindications are limited to only the following:
- Severe allergic reaction (e.g., anaphylaxis) to a dose of DTaP or Tdap or to a vaccine component (Td) is a contraindication to DTaP, Tdap, or Td. Because of the importance of tetanus vaccination, people who experience anaphylactic reactions should be referred to an allergist to determine whether they have a specific allergy to tetanus toxoid and can be desensitized to tetanus toxoid.
- Encephalopathy (e.g., coma, decreased level of consciousness, or prolonged seizures) not attributable to another identifiable cause within 7 days after administration of a previous dose of diphtheria and tetanus toxoids and pertussis vaccine (DTwP, DTaP, or Tdap) is a contraindication to the pertussis component.
Rates of tetanus are currently at a historical low in the United States. Vaccine trials have not previously included tetanus toxoid efficacy, but experts can infer efficacy from protective antitoxin levels. A complete vaccination series likely has a clinical efficacy of virtually 100% for tetanus. A complete series of tetanus vaccination is 4 doses for children younger than 7 years old and 3 doses for people 7 years old or older.
Tetanus Toxoid-Containing Vaccines and Brands* |
Number of Doses |
Recommended Age for All
Doses |
Dose & Route |
| Dose 1 |
Dose 2 |
Dose 3 |
Dose 4 |
Booster |
DAPTACEL® (DTaP)
Sanofi Pasteur |
5 |
6 wks-2 mos |
4 mos |
6 mos |
15-20 mos |
4-6 yrs |
0.5mL
Intramuscular (IM)
|
INFANRIX (DTaP)
Sanofi Pasteur |
5 |
6 wks-2 mos
|
4 mos |
6 mos |
15-20 mos |
4-6 yrs |
0.5mL
IM
|
PEDIARIX (DTaP-HepB-IPV)
GlaxoSmithKline (GSK) |
3 |
6 wks-2 mos |
4 mos |
6 mos |
Not recommended** |
0.5mL
IM
|
Pentacel® (DTaP-IPV/Hib)
Sanofi Pasteur
|
4 |
6 wks-2 mos |
4 mos |
6 mos |
N/A |
15-18 mos |
0.5mL
IM
|
VAXELIS® (DTaP-IPV-Hib-HepB)***
MSP Vaccine Company
|
3 |
6 wks-2 mos |
4 mos |
6 mos† |
N/A |
N/A |
0.5mL
IM |
KINRIX (DTaP-IPV) ††
GlaxoSmithKline (GSK)
|
1-2 |
N/A |
N/A |
N/A |
4-6 yrs†† |
0.5mL
IM
|
Quadracel® (DTaP-IPV) †††
Sanofi Pasteur
|
1-2 |
N/A |
N/A |
N/A |
4 - 6 yrs††† |
0.5mL
IM
|
Adacel® (Tdap) §
Sanofi Pasteur |
Booster |
N/A |
N/A |
N/A |
N/A |
7-64 yrs‡ |
0.5mL
IM
|
Boostrix (Tdap) §
GlaxoSmithKline (GSK)
|
Booster |
N/A |
N/A |
N/A |
N/A |
7-64 yrs‡ |
0.5mL
IM
|
| TENIVAC® (Td) ‡‡ |
3 and/or Booster |
≥7 yrs |
≥7 yrs |
≥7 yrs |
N/A |
≥11 yrs‡‡‡ |
0.5mL
IM
|
*Tetanus-containing vaccines can be administered with other vaccines.
**Because the pertussis antigens contained in INFANRIX and KINRIX are the same as those in PEDIARIX, these children should receive INFANRIX as their fourth dose of DTaP and either INFANRIX or KINRIX as their fifth dose of DTaP. KINRIX or Quadracel® may be used to complete the 4-dose IPV series.
***Preferred for American Indian and Alaska Native infants based on the Haemophilus influenzae type b (Hib) component.
† The recommended minimum age for dose 3 of VAXELIS® is 24 weeks, the minimum age for completion of the hepatitis B vaccine series.
†† KINRIX is approved only for dose 5 of DTaP and dose 4 of IPV.
††† Quadracel® is approved only for dose 5 of DTaP and dose 4 or 5 of IPV.
§ Children 7-9 years old who have never been vaccinated against pertussis, tetanus, or diphtheria should receive a series of three tetanus and diphtheria toxoid-containing vaccines, which includes at least one dose of Tdap.
‡ Either Tdap vaccine administered to a person 65 years old or older is immunogenic and would provide protection; a dose of either vaccine would be considered valid.
‡‡ Primary immunization with TENIVAC® consists of 3 doses, with the first 2 doses administered 2 months apart. The third dose is administered 6-8 months after the second dose.
‡‡‡ TENIVAC® may be used as a booster beginning at 11-12 years of age, and every 10 years thereafter.